Phase 1/2a Study Highlights Potential of CER-0001 in Infantile Epileptic Spasms
Cerecin Inc. has published encouraging early clinical data for CER-0001 (tricaprilin), an investigational ketogenic drug being developed for rare and severe forms of epilepsy.
The Phase 1/2a study showed that CER-0001 was generally well tolerated and demonstrated seizure reduction in infants with drug-resistant infantile epileptic spasms.
Study Publication and Scope
The study, titled “Ketogenic drug tricaprilin (CER-0001) for the treatment of refractory infantile epileptic spasms: a phase 1/2a study,” was published on February 20, 2026 in Frontiers in Pediatrics.
The results support the continued clinical development of CER-0001 as a metabolic therapy for developmental and epileptic encephalopathies (DEE).
Understanding Infantile Epileptic Spasms
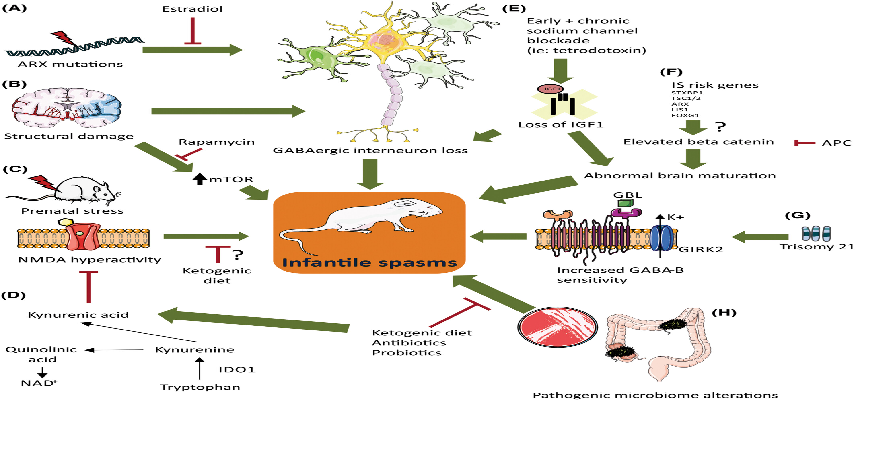
Infantile epileptic spasms, also known as West syndrome, are a rare and devastating epilepsy that typically begins between 3 and 24 months of age.
Key challenges include:
- High seizure burden in early life
- Significant risk of developmental delay
- Long-term neurological disability
Despite existing treatments, 40–60% of patients do not respond adequately to first-line therapies, underscoring the need for new approaches.
A Drug-Based Alternative to the Ketogenic Diet
CER-0001 is designed to deliver the benefits of therapeutic ketosis without requiring strict dietary adherence.
Unlike traditional ketogenic diets:
- CER-0001 is administered as an oral prescription drug
- It aims to simplify metabolic therapy for families and clinicians
- It may expand access to ketosis-based treatment strategies
This positions CER-0001 as a practical alternative to diet-based metabolic interventions.
Promising Early Clinical Results
The open-label Phase 1/2a study enrolled eight infants with refractory epileptic spasms across clinical sites in Australia and Singapore.
Key takeaways:
- CER-0001 was generally well tolerated
- Early signals of seizure reduction were observed
- The therapy avoided the burden of strict dietary intervention
These findings suggest a novel pharmacological approach to ketogenic therapy in rare pediatric epilepsies.
Next Steps in Development
Based on the Phase 1/2a results, Cerecin plans to advance CER-0001 into a larger randomized controlled study. The next trial will aim to:
- Confirm efficacy in a broader patient population
- Further evaluate safety and dosing
- Support development in developmental and epileptic encephalopathies
Broader Commitment to Rare Neurological Diseases
Cerecin is focused on developing metabolic medicines that address underlying energy deficits in neurological disorders. In addition to rare epilepsies, CER-0001 is also being explored for:
- Alzheimer’s disease
- Migraine
CER-0001 remains an investigational therapy and has not been approved by any regulatory authority.
The Bigger Picture
For families affected by infantile epileptic spasms, treatment options remain limited and challenging. This early study suggests that CER-0001 could represent a more accessible metabolic therapy, combining the biological benefits of ketosis with the practicality of a prescription drug—an important step forward in rare epilepsy research.

Optimize Your trial insights with Clival Database.
Are you exhausted from the uncertainty of trial insights pricing? Clival Database ensures the clarity in the midst of the global scenario for clinical trials to you.Clival Database is one of the best databases that offers an outstanding number of clinical trial data in terms of 50,000+ molecules and from primary regulatory markets as well as new entrants like Indian and Chinese markets.
With Clival, you get accurate positioning of historical sales data, patent database, company profiling, safety & efficacy, and prediction of launch of new innovative molecules helping you to align your research and driving down the cost.
To add value, we further break down our analytics for you so that improving your operational effectiveness; optimizing your clinical trials; and offering you accurate and high-quality data at lowest possible prices becomes possible.
Elevate your trial success rate with the cutting-edge insights from Clival database.
Check it out today and make more informed sourcing decisions! Learn More!